 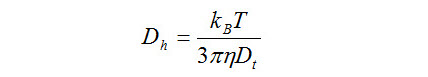
It is well understood that the process of aggregation towards amyloid fibrils from a monomeric state is a nucleation-dependant mechanism, analogous to crystal growth. Due to their significance in human pathologies, there have been unprecedented efforts towards physiochemical understanding of aggregation and amyloid formation over the last two decades. In all amyloid diseases, such protein aggregates have been implicated fully or partly, in the etiology of the disease. A biochemical commonality in these diseases is that the protein involved forms pathogenic aggregates, irrespective of whether the monomeric protein is in the misfolded or intrinsically disordered forms, that have a consensus structural moiety, commonly known as “amyloids”. The modification we are reporting is not only useful in A β aggregation but also will be important for accurate measurements in all protein aggregation systems.Īberrant misfolding and aggregation of proteins have been implicated in over 40 different human pathologies including Alzheimer’s disease (AD), Parkinson’s disease, type 2 diabetes, transmissible spongiform encephalopathies (TSE) or Prion diseases, Huntington’s disease and Cruedfelt-Jacob disease (CJD). 
Our modifications also involve inclusion of improved shape factors of molecules and more appropriate viscosities. We argue that the standard Stokes-Einstein’s equation is insufficient to understand the temporal variations in diffusion when trying to understand the aggregation behavior of A β 42 proteins. Comparison with experiments for variations in diffusion coefficients over time reveals very similar trends. The net result is that the diffusion coefficient which is inversely proportional to the viscosity of the system is now dependent on the concentration of the different molecules as well as their proper shapes. The novelty of our model is that in addition to the volume fractions, it incorporates previously published reports on the dimensions of the protofibrils and their aggregates to formulate a more appropriate shape rather than mere spheres. This effective viscosity is modeled as a function of the volume fractions of the different species of molecules. The modified Stokes-Einstein equation incorporates an effective viscosity for the mixture consisting of the macromolecules and solvent where the lateral association reaction occurs. Specifically, we have focused on validating this model in protofibrill lateral association reactions along the aggregation pathway, which has been experimentally well characterized. 
Here, we report a modification in the classical Stokes-Einstein equation using a mixture theory approach, in order to accommodate the changes in viscosity of the solvent due to the changes in solute size and shape, to implement a more realistic model for A β aggregation involved in Alzheimer’s disease. An important relation from which hydrodynamic radii of the aggregate is routinely measured is the classic Stokes-Einstein equation. In all amyloid diseases, protein aggregates have been implicated fully or partly, in the etiology of the disease. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
